Spine Surgery Instrumentation: Everything You Need to Know
Spine surgery instrumentation is the full complement of implantable hardware, fixation devices, cutting instruments, and delivery systems used to stabilize, decompress, fuse, or replace structures of the spinal column. From titanium pedicle screw constructs to expandable interbody cages to cervical plating systems, these instruments define what a spine surgeon can accomplish in the operating room — and what outcomes the patient takes home.
This guide covers every major category of spine instrumentation in use today: posterior fixation systems, interbody fusion cages across all four surgical corridors, cervical plating, artificial disc replacement, minimally invasive platforms, biologics that drive fusion, and the navigation and robotic technologies reshaping how these devices get placed. It is written for spine surgeons, surgical teams, device representatives, and procurement professionals who need to understand what is available, what matters clinically, and how to source it without supply chain delays.
Overview of Spine Surgery Instrumentation
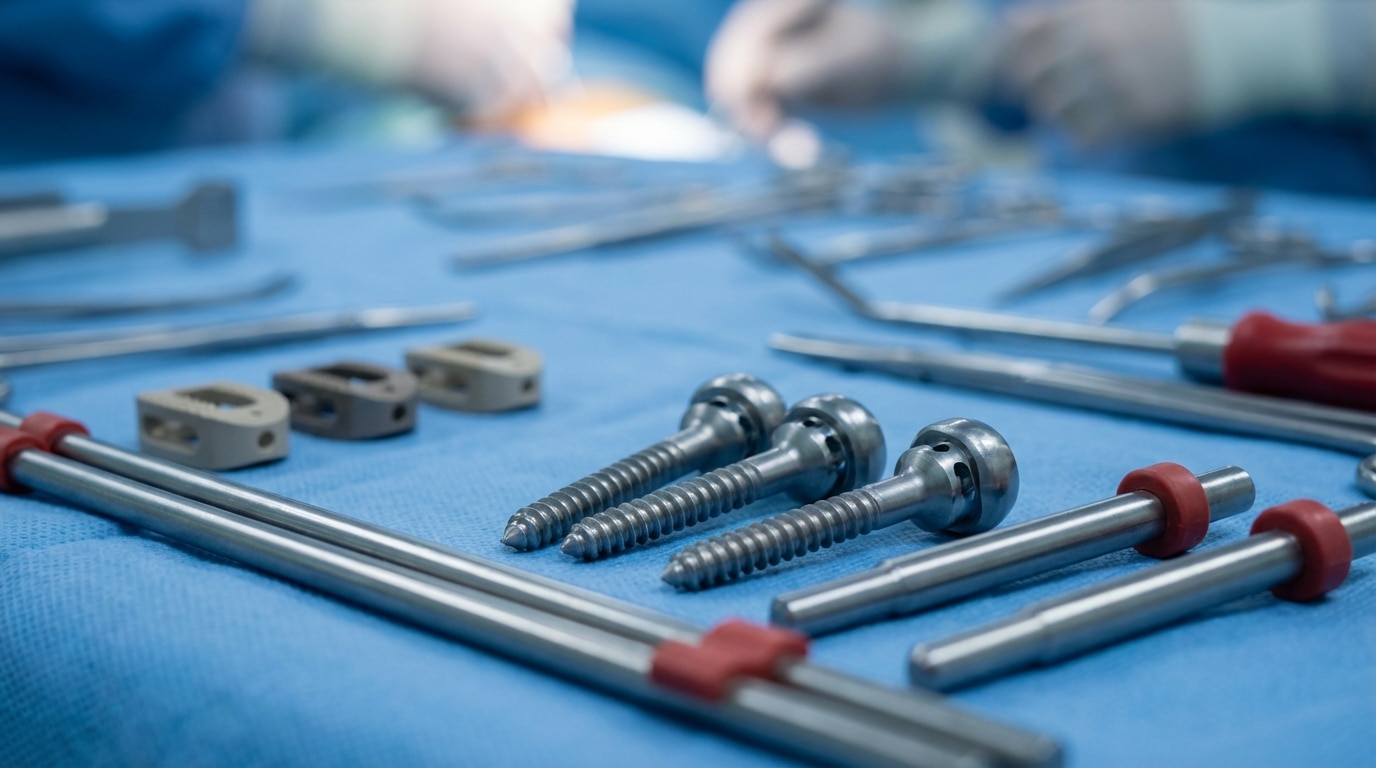
Spine surgery instrumentation falls into three broad functional categories: fixation hardware (pedicle screws, rods, plates, hooks, and wires that stabilize the spine), interbody devices (cages and spacers that restore disc height and promote fusion between vertebral bodies), and access and delivery instruments (retractors, dilators, cannulas, and specialized tools that create the surgical corridor and seat the implants).
Every spine procedure — whether a single-level anterior cervical discectomy and fusion (ACDF) or a multi-level posterior lumbar interbody fusion with deformity correction — requires a specific tray set configured for that approach, that anatomy, and that patient. A complete instrumentation set for a posterior lumbar fusion, for example, typically includes:
- Pedicle access instruments (awls, probes, taps, pedicle finders)
- Polyaxial or monoaxial pedicle screws in multiple diameters (5.5mm through 8.5mm) and lengths (30mm through 60mm)
- Titanium or cobalt-chrome rods (pre-contoured or straight, 5.5mm or 6.0mm diameter)
- Rod reduction instruments and set screws
- Interbody cage trials and implants
- Cage insertion instruments
- Cross-connectors (for multi-level constructs)
- Bone graft preparation instruments
The surgical supply chain for spine instrumentation is distinct from general orthopedics. Spine cases are instrument-intensive, approach-specific, and frequently scheduled with 24 to 48 hours of lead time. When the tray set does not show up, the case does not happen. This makes supplier reliability and inventory availability the two most consequential variables in spine device procurement — more on that in the supplier selection section below.
Materials in Modern Spine Implants
Most posterior fixation hardware is manufactured from Ti-6Al-4V titanium alloy (Grade 5 titanium), which provides high fatigue strength with excellent biocompatibility and MRI compatibility. Cobalt-chrome (CoCr) alloys are used in some rod systems where higher stiffness is desired for deformity correction. Interbody cages are manufactured from PEEK (polyether ether ketone), titanium, porous tantalum, or 3D-printed titanium with trabecular surface textures designed to promote bony on-growth.
The trend toward 3D-printed titanium interbody cages has been significant. These devices use additive manufacturing to create open-lattice structures that mimic cancellous bone porosity, theoretically improving osseointegration while maintaining the structural stiffness needed to bear axial loads.
Pedicle Screw Systems
Pedicle screw fixation is the workhorse of posterior spine surgery. The concept is straightforward: a screw is placed through the pedicle of the vertebra into the vertebral body, and bilateral screws at adjacent levels are connected by rods to create a rigid or semi-rigid construct that immobilizes the instrumented segment while fusion consolidates.
Modern pedicle screw systems have evolved substantially from the original Cotrel-Dubousset instrumentation of the 1980s. Current systems offer several screw head configurations:
- Polyaxial screws — the screw head allows multi-directional angulation (typically 25-30 degrees of freedom) before the rod is locked in place, making rod-screw coupling easier across multiple levels and accommodating anatomic variability
- Monoaxial screws — fixed-head design providing maximum rigidity, used primarily in fracture fixation and deformity constructs where controlled correction forces are applied
- Uniplanar screws — allow angulation in one plane only, offering a middle ground between polyaxial flexibility and monoaxial rigidity
- Reduction screws — extended-tab polyaxial screws that allow sequential rod reduction in cases with spondylolisthesis or kyphotic deformity, bringing the rod down to the screw rather than forcing the anatomy to the rod
Cannulated vs. Solid Screws
Cannulated pedicle screws have a hollow core that allows them to be placed over a guidewire under fluoroscopic guidance. This is the standard technique in minimally invasive surgery (MIS) and percutaneous screw placement. Solid-core screws are the traditional design, offering theoretically higher pullout strength and are the standard in open posterior approaches.
Screw Augmentation
In patients with osteoporotic bone — increasingly common as the spine surgery population ages — screw pullout is a real clinical problem. Cement augmentation using PMMA (polymethylmethacrylate) injected through cannulated or fenestrated screws can increase pullout strength by 100-200%. Fenestrated screws have side holes along the shaft that allow PMMA to extrude into the cancellous bone of the vertebral body, creating a cement mantle around the screw.
Expandable pedicle screws are another option. These screws have a mechanism at the tip that expands after insertion, increasing the screw’s purchase in the vertebral body without the complications associated with cement (leakage, embolism, thermal necrosis).
Rod Options
Rods connect the pedicle screws and define the biomechanical behavior of the construct. Standard options include:
| Rod Material | Diameter | Stiffness | Primary Use |
|---|---|---|---|
| Titanium alloy | 5.5mm / 6.0mm | Moderate | Standard degenerative cases, short constructs |
| Cobalt-chrome | 5.5mm / 6.0mm | High | Deformity correction, long constructs |
| PEEK rods | 6.0mm | Low (semi-rigid) | Dynamic stabilization, motion preservation adjacent to fusion |
| Pre-contoured rods | Varies | Varies | Reduce intraoperative rod bending time, maintain sagittal alignment |
Rod contouring is a critical intraoperative step. The rod must match the patient’s sagittal profile — specifically the lumbar lordosis and thoracic kyphosis targets determined by preoperative planning. French benders, in-situ benders, and pre-contoured rods all address this need, but improper contouring remains a source of sagittal imbalance and proximal junctional failure.
Interbody Fusion Cages: ALIF, PLIF, TLIF, and LLIF
Interbody fusion cages are implanted into the disc space after discectomy to restore disc height, provide structural support, and create a contained environment for bone graft to achieve solid arthrodesis. The surgical approach determines the cage geometry, insertion technique, and biomechanical profile.
Comparison of Interbody Fusion Approaches
| Feature | ALIF | PLIF | TLIF | LLIF (XLIF/DLIF) |
|---|---|---|---|---|
| Approach | Anterior (retroperitoneal or transperitoneal) | Posterior (bilateral laminotomy) | Posterior (unilateral transforaminal) | Lateral (transpsoas or anterior-to-psoas) |
| Levels treated | L4-S1 most common | L1-S1 | L1-S1 | L1-L5 (not L5-S1) |
| Cage size | Large footprint (26-40mm wide) | Small paired cages (8-12mm wide) | Medium banana/crescent (10-14mm wide) | Large footprint (18-22mm wide, 45-60mm long) |
| Lordosis restoration | Excellent (up to 20-30 degrees with hyperlordotic cages) | Moderate | Moderate to good | Good (10-20 degrees with lordotic cages) |
| Neural retraction | None | Bilateral thecal sac retraction | Unilateral nerve root retraction | None (but lumbar plexus at risk) |
| Standalone capable | Yes (with integrated fixation) | No (requires posterior fixation) | No (requires posterior fixation) | Yes at some levels (with integrated fixation) or supplemental posterior fixation |
| Vascular risk | Highest (great vessels) | Low | Low | Low |
| Access surgeon needed | Yes (vascular or general surgeon) | No | No | No (but neuromonitoring required) |
ALIF (Anterior Lumbar Interbody Fusion)
ALIF cages provide the largest possible footprint on the vertebral endplate because the anterior approach allows direct, unobstructed access to the disc space without neural structures in the way. This large footprint translates to better load distribution, higher resistance to subsidence, and the ability to use hyperlordotic cage designs (15, 20, or 30 degrees) that restore segmental and regional lumbar lordosis.
Modern ALIF cages often include integrated fixation — either built-in screws that anchor through the cage into the adjacent vertebral bodies, or a separate anterior plate. Some designs combine both. Standalone ALIF at L5-S1 (where the iliac crests provide natural lateral restraint) has shown solid fusion rates in the literature, reducing the need for a second posterior procedure in appropriate patients.
PLIF (Posterior Lumbar Interbody Fusion)
PLIF uses a posterior midline approach with bilateral laminotomies to access the disc space. Two smaller cages are placed side by side. The primary advantage is that the spine surgeon can perform decompression, cage placement, and posterior fixation through a single posterior exposure. The primary disadvantage is the need to retract the thecal sac and bilateral nerve roots, which carries a higher risk of dural tear and nerve injury compared to TLIF.
TLIF (Transforaminal Lumbar Interbody Fusion)
TLIF has largely replaced PLIF at many spine centers. The transforaminal approach accesses the disc space through the neural foramen from one side, requiring retraction of only the traversing nerve root on the approach side. A single, typically banana-shaped or crescent-shaped cage is inserted obliquely across the disc space. TLIF is well-suited to MIS techniques using tubular retractors and is the most commonly performed interbody fusion approach in the United States.
LLIF (Lateral Lumbar Interbody Fusion)
Lateral approaches — XLIF (eXtreme Lateral Interbody Fusion), DLIF (Direct Lateral), and ATP (Anterior to Psoas) — access the disc space through a flank incision, passing through or anterior to the psoas muscle. These approaches avoid the posterior musculature entirely and provide a large cage footprint similar to ALIF. The primary concern is the lumbar plexus, which runs through the psoas muscle, making intraoperative neuromonitoring (EMG) mandatory.
LLIF cannot reliably access L5-S1 due to the iliac crest, so treatment of the lumbosacral junction requires either an ALIF or TLIF at that level in a staged or same-day combined approach.
Expandable Cages
A significant development in interbody technology is the expandable cage. These devices are inserted in a collapsed profile — critical for MIS approaches where the working corridor is narrow — and then expanded in situ to the desired height. Expandable TLIF and LLIF cages allow the surgeon to trial and adjust disc height and lordosis after the cage is seated, which is not possible with static (fixed-height) implants. Some expandable cages also incorporate articulating endplates that conform to the vertebral endplate geometry, potentially reducing the risk of subsidence.
Cervical Plating and Fixation
Anterior cervical discectomy and fusion (ACDF) is one of the most commonly performed spine procedures worldwide, and the instrumentation is distinct from lumbar systems. After discectomy and interbody graft or cage placement, an anterior cervical plate is applied across the fused level(s) to provide immediate stability, prevent graft extrusion, and improve fusion rates.
Anterior Cervical Plates
Modern anterior cervical plates are low-profile titanium constructs ranging from single-level (approximately 20-24mm in length) to multi-level designs. Key design features include:
- Variable-angle screws — allow 15-25 degrees of angulation, giving the surgeon flexibility in screw trajectory to avoid adjacent disc spaces
- Locking mechanisms — prevent screw backout, which was a common failure mode with earlier plate designs. Current plates use either a rotating cam lock, a secondary locking screw at each hole, or a deformable washer mechanism
- Dynamic vs. constrained designs — dynamic plates allow controlled settling (1-2mm) as the graft incorporates, theoretically maintaining graft loading and promoting fusion. Constrained (rigid) plates maintain fixed graft height
- Zero-profile integrated devices — combine the interbody cage and the anterior fixation into a single implant that sits entirely within the disc space, avoiding the plate-on-bone profile that can cause dysphagia. These devices use integrated screws or anchors that lock into the vertebral body from within the disc space
Posterior Cervical Fixation
Posterior cervical instrumentation is used for multilevel fusions, fractures, instability, and deformity. Options include:
- Lateral mass screws — placed into the lateral mass of C3-C6, the workhorse of subaxial posterior cervical fixation
- Cervical pedicle screws — provide the strongest fixation but carry higher risk due to the vertebral artery and spinal cord proximity. Used selectively at C2, C7, and in revision or deformity cases
- C1-C2 fixation (Goel-Harms technique) — C1 lateral mass screws paired with C2 pedicle or pars screws, connected by rods. The standard technique for atlantoaxial instability
- Occipitocervical fixation — a plate or rod construct anchored to the occiput with bicortical screws, extending to the subaxial cervical or upper thoracic spine. Used for craniovertebral junction instability
Artificial Disc Replacement
Total disc replacement (TDR) is a motion-preserving alternative to fusion for select patients with symptomatic disc degeneration. The concept: rather than eliminating motion at the diseased segment, replace the disc with an articulating prosthesis that maintains physiologic kinematics.
Cervical Disc Replacement
Cervical TDR has accumulated the strongest evidence base and the broadest FDA approval among disc replacement technologies. Multiple devices are FDA-approved for single and two-level use in the cervical spine, and long-term data (10+ year follow-up from IDE trials) have demonstrated non-inferiority or superiority to ACDF for the approved indications, with lower rates of adjacent segment disease requiring reoperation.
Current cervical disc designs fall into several categories:
- Metal-on-polymer — cobalt-chrome endplates articulating on a UHMWPE (ultra-high molecular weight polyethylene) core. Examples: Prestige LP (metal-on-metal), ProDisc-C (metal-on-poly), Mobi-C (mobile-bearing metal-on-poly)
- Metal-on-metal — CoCr articulation surfaces
- Compressible core designs — deformable polymer core that allows both rotation and axial compression, more closely mimicking the native disc’s viscoelastic behavior
Lumbar Disc Replacement
Lumbar TDR is less widely adopted than cervical. It is FDA-approved for single-level use at L3-S1 through an anterior approach (same exposure as ALIF). Patient selection is critical and more restrictive than in the cervical spine: ideal candidates have single-level disease, no significant facet arthrosis, no instability, and no posterior element pathology. The ProDisc-L and activL are among the FDA-approved lumbar TDR devices.
Clinical Considerations
Disc replacement is not appropriate for patients with osteoporosis, spinal stenosis requiring posterior decompression, spondylolisthesis, facet arthrosis, or prior surgery at the index level. The implant must sit on strong endplate bone and function in a biomechanically intact posterior column. Patient selection accounts for the majority of outcome variance in TDR.
Minimally Invasive Spine Instrumentation
Minimally invasive spine surgery (MIS) uses smaller incisions, tubular retractors, and specialized instruments to accomplish the same surgical objectives as open approaches — decompression, fusion, and fixation — while preserving the posterior musculature, reducing blood loss, and shortening hospital stay.
MIS instrumentation differs from open instrumentation in several key ways:
Open vs. Minimally Invasive Instrumentation
| Feature | Open Surgery | MIS Surgery |
|---|---|---|
| Exposure | Direct visualization, wide exposure | Tubular retractor, limited direct visualization |
| Screw placement | Freehand with anatomic landmarks | Percutaneous, fluoroscopy- or navigation-guided |
| Screw type | Standard top-loading | Cannulated, percutaneous with extender tabs |
| Rod insertion | Direct placement and contouring | Subfascial rod passage through screw extenders |
| Cage delivery | Direct insertion under visualization | Through tubular retractor with specialized inserters |
| Muscle damage | Subperiosteal dissection, denervation of multifidus | Muscle-splitting approach through Wiltse interval |
| Blood loss | Higher | Significantly lower |
| Hospital stay | 2-4 days typical | 0-2 days typical |
| Radiation exposure | Lower (less fluoro) | Higher (more fluoro, unless using navigation) |
| Learning curve | Standard residency training | Fellowship-level, case volume dependent |
Key MIS Instrument Categories
Tubular retractor systems are the foundation of MIS spine surgery. Sequential dilators spread the paraspinal muscles rather than stripping them, and a tubular retractor (typically 18-26mm diameter) holds the working corridor open. The surgeon operates through this tube using long-handled instruments, a headlight or tubular light source, and microscope or endoscope visualization.
Percutaneous pedicle screw systems use a Jamshidi needle inserted under fluoroscopy to access the pedicle, followed by a guidewire, cannulated tap, and cannulated screw. The screw has an extender tab that projects above the skin, allowing the surgeon to pass a rod subfascially through all the screw extenders and lock it in place without ever exposing the muscle or fascia.
Endoscopic spine systems represent the newest frontier. Full-endoscopic transforaminal or interlaminar discectomy uses a 7-8mm working channel endoscope under continuous saline irrigation. Some centers are now performing endoscopic-assisted interbody fusion. These systems require specialized training and are not yet standard at most institutions, but they represent the direction of the field.
Biologics in Spine Surgery
Hardware creates stability. Biologics create fusion. No spinal fusion construct is complete without a biological strategy to achieve bony arthrodesis across the instrumented segments. The choice of biologic graft material is as consequential as the choice of hardware — a technically perfect construct will fail if fusion does not consolidate.
Autograft
Local autograft (bone harvested from the laminectomy or facetectomy during the decompression) remains the gold standard biological material. It contains all three elements of the “diamond concept” of bone healing: osteogenic cells (viable osteoblasts and osteoprogenitor cells), osteoinductive signals (BMPs, growth factors in the bone matrix), and an osteoconductive scaffold (the cancellous bone architecture itself). Iliac crest bone graft (ICBG) provides a larger volume of autograft but adds donor site morbidity — pain, hematoma, and sensory loss at the harvest site.
Allograft
Structural allograft (cadaveric bone) is used as a spacer in anterior cervical fusions (femoral ring allograft or machined cortical allograft) and as cancellous chips to supplement local autograft in posterolateral fusion beds. Allograft is osteoconductive and weakly osteoinductive but not osteogenic — the processing eliminates viable cells.
Bone Morphogenetic Protein (BMP)
Recombinant human BMP-2 (rhBMP-2, marketed as INFUSE by Medtronic) is the most potent osteoinductive agent commercially available. It is FDA-approved for use in ALIF with a specific cage at a specific dose. Off-label use in posterior lumbar fusion and cervical spine has been associated with significant complications — including ectopic bone formation, radiculitis, osteolysis, and airway swelling in the cervical spine — and its use should follow evidence-based protocols with appropriate informed consent. The FDA safety communication on BMP complications provides critical guidance for spine surgeons considering its use.
Demineralized Bone Matrix (DBM)
DBM products are processed allograft in which the mineral content has been removed, exposing the collagen matrix and endogenous growth factors (including low concentrations of native BMPs). Available as putty, gel, paste, strips, and fiber forms, DBM is widely used as a graft extender — mixed with local autograft to increase graft volume without increasing harvest morbidity.
Cellular Therapies and Stem Cell Products
Cellular allograft products containing viable mesenchymal stem cells (MSCs), osteoprogenitor cells, or concentrated bone marrow aspirate (BMA) represent a growing segment of the spine biologics market. These products aim to add the osteogenic component that standard allograft and DBM lack. Products containing viable cells from amniotic tissue, umbilical cord tissue, and placental tissue are marketed under the FDA’s 361 HCT/P regulatory pathway.
The evidence base for these cellular products is still developing. Surgeons should evaluate the published clinical data for any cellular biologic before adoption. That said, combining a cellular allograft with local autograft and an osteoconductive scaffold (ceramic granules, for example) creates a composite graft strategy that addresses all three pillars of the biological fusion environment.
Synthetics and Ceramics
Synthetic bone graft substitutes — including beta-tricalcium phosphate (TCP), hydroxyapatite (HA), and biphasic calcium phosphate ceramics — serve as osteoconductive scaffolds. They are most commonly used as graft extenders in posterolateral fusion or to fill interbody cages in combination with more biologically active materials. They do not have osteoinductive or osteogenic properties on their own.
Navigation and Robotics in Spine Surgery
Intraoperative navigation and robotic-assisted surgery have changed the accuracy and safety profile of spine instrumentation placement. These technologies address a fundamental challenge in spine surgery: pedicle screws, cervical lateral mass screws, and sacral alar-iliac screws all pass through narrow bony corridors in close proximity to the spinal cord, nerve roots, and major vessels. Misplacement carries real consequences.
Intraoperative Navigation
Computer-assisted navigation uses either a preoperative CT scan (registered to the patient’s anatomy intraoperatively) or an intraoperative 3D spin (O-arm, Airo, or cone-beam CT) to create a real-time three-dimensional map of the patient’s spine. Tracked instruments and implants are displayed on this map, giving the surgeon continuous visual feedback on trajectory, depth, and position during screw placement.
Published data consistently demonstrate that navigation reduces pedicle screw misplacement rates compared to freehand technique. A meta-analysis published in Spine found that navigation reduced the rate of pedicle cortex breach from approximately 15% (freehand) to under 6% (navigated), with the most significant improvements in the thoracic spine where pedicle anatomy is most challenging (Tian NF et al., Spine, 2011).
Robotic-Assisted Spine Surgery
Robotic platforms add a mechanical guidance component to navigation. After the surgeon plans screw trajectories on preoperative or intraoperative imaging, the robotic arm positions a drill guide at the planned entry point and trajectory. The surgeon then drills and places the screw through the guide, constrained to the planned path.
Current FDA-cleared robotic platforms for spine surgery include Globus Medical’s ExcelsiusGPS, Medtronic’s Mazor X Stealth Edition, NuVasive’s Pulse platform, and Zimmer Biomet’s ROSA ONE Spine. These systems differ in their integration with navigation, their workflow (CT-to-plan vs. intraoperative registration), and their robotic architecture, but all share the core function of guided screw placement.
The clinical data on robotics is encouraging but still evolving. Accuracy rates for robotic pedicle screw placement consistently exceed 95% in published series. The larger question — whether improved accuracy translates to better clinical outcomes and fewer reoperations — requires longer follow-up and larger comparative studies. The North American Spine Society (NASS) continues to track outcomes data on navigation and robotic technologies.
Augmented Reality
Augmented reality (AR) systems project navigational data directly into the surgeon’s field of view through a headset or heads-up display, eliminating the need to look away from the surgical field to a separate navigation screen. Several AR navigation platforms are in clinical use or advanced development for spine surgery. Early data suggest comparable accuracy to standard navigation with improved surgical workflow and reduced need to break sterile field to reference the screen.
Choosing a Spine Device Supplier
Spine instrumentation procurement is not a commodity transaction. The wrong supplier does not just deliver inferior products — it delivers late, delivers incomplete tray sets, fails to provide qualified technical support in the OR, and creates case cancellations that cost the facility revenue and the patient time.
When evaluating a spine device supplier, surgical facilities and procurement teams should assess the following:
Inventory Availability and Lead Time
This is the single most important operational factor. Spine cases are frequently booked with short lead times and require specific tray configurations (screw sizes, cage options, rod lengths, biologics) matched to the individual patient’s anatomy and the surgeon’s plan. A supplier that operates from fully stocked warehouses with zero-lead-time processing eliminates the risk of case delays and cancellations caused by supply chain gaps.
Ask any prospective supplier: If I book a multi-level posterior lumbar fusion with interbody cages for Wednesday morning, when does the complete tray set arrive at the facility? The answer tells you everything about their operational model.
Product Portfolio Breadth
A supplier that covers pedicle screw systems, interbody cages (all approaches), cervical plating, biologics, and MIS instrumentation from a single source simplifies procurement, reduces vendor credentialing complexity, and ensures compatibility across product lines. Multi-vendor sourcing is sometimes necessary but always adds logistical friction.
Technical Support and Field Coverage
Spine cases require a knowledgeable device representative or surgical technologist in the room who understands the instrumentation, the surgical technique, and the implant selection process. This person manages the tray set, ensures the correct implants are available, and provides real-time technical support to the surgical team. The quality of this field support varies dramatically between suppliers.
Regulatory Compliance
All spine implants must be FDA-cleared (510(k) or PMA), and the supplier must maintain proper documentation, lot traceability, and quality management systems. Facilities should verify that their supplier operates under a compliant quality system and can provide certificates of conformity, device history records, and recall/field safety notice communication.
Pricing and Contracting
Spine implant pricing has historically been opaque. GPO (Group Purchasing Organization) contracts, direct agreements, and ASC-specific pricing models all exist. A good supplier offers transparent, competitive pricing with clear per-implant costs and no hidden fees for tray management, loaner sets, or instrument processing.
SLR Medical Consulting supplies orthopedic and spine hardware, biologics, and sports medicine instrumentation to surgical facilities nationwide. With fully stocked warehouses and zero-lead-time processing, SLR eliminates the supply chain uncertainty that disrupts surgical schedules. Browse our spine hardware catalog or place a surgical order to discuss your facility’s instrumentation needs.
Frequently Asked Questions About Spine Surgery Instrumentation
What is included in a standard spine surgery instrumentation set?
A standard posterior lumbar fusion instrumentation set includes pedicle access instruments (awls, probes, and taps), pedicle screws in multiple diameters and lengths, connecting rods, set screws, rod reduction instruments, and cross-connectors. If interbody fusion is planned, the set also includes disc preparation instruments, cage trials, the interbody cage implant, and cage insertion tools. Biologics (bone graft materials) are supplied separately. The specific configuration varies by surgical approach, number of levels, and surgeon preference.
What is the difference between titanium and PEEK interbody cages?
Titanium cages (including 3D-printed titanium) offer superior osseointegration — bone grows directly into the porous titanium surface, creating a biological bond between the implant and the vertebral endplate. PEEK cages have a modulus of elasticity closer to bone, which may reduce stress shielding and subsidence, and they do not produce artifact on postoperative CT or MRI, making radiographic fusion assessment easier. Both materials have strong clinical track records. The choice often depends on surgeon preference, specific clinical scenario (e.g., osteoporotic bone may favor titanium for better integration), and whether postoperative imaging clarity is a priority.
How do minimally invasive spine instruments differ from open instruments?
MIS instruments are designed to work through narrow corridors — typically 18-26mm tubular retractors or percutaneous access. This means longer instrument shafts, smaller working ends, cannulated designs for guidewire-directed placement, and specialized visualization tools (endoscopes, tubular retractor-compatible microscope adapters). Percutaneous pedicle screw systems use extender tabs that project above the skin surface, allowing the surgeon to manipulate and lock the screw without direct muscle exposure. Open instruments are shorter, wider, and designed for use under direct visualization with standard retraction.
What biologics are most commonly used in spine fusion surgery?
Local autograft — bone harvested during the decompression portion of the procedure — is the most commonly used primary graft material. It is typically supplemented with a graft extender such as demineralized bone matrix (DBM) or a ceramic scaffold to increase graft volume. For anterior fusions, structural allograft or the interbody cage itself provides the mechanical scaffold, with DBM or cellular allograft packed inside. Recombinant BMP-2 is used in specific clinical scenarios, primarily FDA-approved for ALIF, with off-label posterior use declining due to complication concerns. Cellular allografts containing viable stem cells or osteoprogenitor cells are an increasingly adopted option for supplementing fusion biology.
How accurate is robotic-assisted pedicle screw placement?
Published clinical series report accuracy rates of 95-99% for robotic-assisted pedicle screw placement, defined as screws entirely within the pedicle cortex (Gertzbein-Robbins Grade A) or with less than 2mm of breach (Grade B). This compares to approximately 85-92% accuracy for freehand placement in most published literature. The improvement is most significant in the thoracic spine, deformity cases, and revision surgery where anatomy is distorted. However, robotic guidance does not eliminate all malpositioned screws, and the surgeon must always verify screw position intraoperatively regardless of the technology used.
What should a surgical facility look for in a spine device supplier?
The five critical evaluation criteria are: (1) inventory availability and delivery lead time — can they deliver complete tray sets reliably within your booking window; (2) product portfolio breadth — do they cover screws, cages, plates, biologics, and MIS instruments from a single source; (3) field technical support quality — is a knowledgeable representative available for case coverage; (4) regulatory compliance and traceability documentation; and (5) transparent, competitive pricing. The most costly supplier failure is not an overpriced implant — it is a cancelled case due to a missing tray set. Prioritize operational reliability above all else.
About SLR Medical Consulting: SLR Medical Consulting has been supplying surgical facilities nationwide for over a decade with orthopedic hardware, spine instrumentation, biologics, and sports medicine devices. Our zero-lead-time delivery model means your surgical schedule runs on your timeline, not your supply chain’s. Explore our hardware catalog or place a surgical order today.